JUVÉDERM
WHAT IS JUVEDERM?
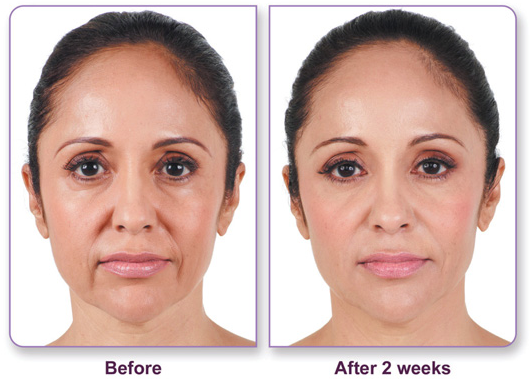
JUVÉDERM Ultra and JUVÉDERM Ultra Plus, approved by the FDA in June 2006, are the ‘next generation’ of hyaluronic acid (HA) dermal fillers and are the only hyaluronic acid dermal fillers FDA-approved to last up to one year, providing a smooth, long-lasting correction of moderate to severe facial wrinkles and folds. The JUVÉDERM dermal filler family of products is the latest advance in HA dermal filler options from Allergan, Inc.
JUVÉDERM Ultra and JUVÉDERM Ultra Plus are the only hyaluronic acid dermal fillers developed using the proprietary HYLACROSS technology, a technologically advanced manufacturing process that results in a malleable, smooth gel that flows easily into the skin and creates a smooth, natural look and feel.
JUVÉDERM dermal fillers are natural, biodegradable and were the first hyaluronic acid dermal fillers to demonstrate safety and effectiveness in patients of color. Studies with JUVÉDERM demonstrated no increased risk of hyperpigmentation or hypertrophic scarring in patients of color.
HOW JUVEDERM WORKS
The key component in JUVÉDERM, hyaluronic acid, is a naturally occurring, biodegradable complex sugar found in the human body and in all living animals. Among other things, hyaluronic acid hydrates the skin and adds volume, contributing to the overall appearance of the skin.
The ability of cells to produce hyaluronic acid diminishes with age, often resulting in the formation of facial wrinkles and folds as the skin loses volume. JUVÉDERM is administered in small doses by intradermal injection directly into moderate to severe facial wrinkles and folds, such as nasolabial folds (the “parentheses” along the side of the nose and mouth), temporarily filling and augmenting the treated area.
WHAT TO EXPECT DURING THE TREATMENT
During a JUVÉDERM® treatment, the area to be injected will be cleansed, usually with alcohol swabs, and no anesthetic is necessary. Clients can request a topical anesthetic cream for comfort. The physician may request that specific expressions be made to identify facial muscle contractions and mark near the injection sites with temporary ink. A series of injections are then administered under the skin using a fine needle to augment, or fill, the soft tissue of the dermis to add volume and diminish wrinkles and folds on the surface of the skin.
A typical JUVÉDERM® treatment takes approximately 10-15 minutes, and clients usually return to normal activities directly after the injections. No allergy skin tests are required.
SIDE EFFECTS
The most commonly reported side effects are temporary injection site redness, swelling, pain/ tenderness, firmness, swelling, lumps/bumps, and bruising. Exposure of the treated area to excessive sun, and extreme cold weather should be minimized until any initial swelling and redness has resolved. If laser treatment, chemical peel or any other procedure based on active dermal response is considered after treatment with JUVÉDERM injectable gel, there is a possible risk of an inflammatory reaction at the treatment site.
Patients who are using substances that can prolong bleeding, such as aspirin or ibuprofen, as with any injection, may experience increased bruising or bleeding at injection site. You should inform your physician before treatment if you are using these types of substances. As with all skin injection procedures there is a risk of infection. JUVÉDERM injectable gel should be used with caution in patients on immunosuppressive therapy, or therapy used to decrease the body’s immune response, as there may be an increased risk of infection. The safety of JUVÉDERM injectable gel in patients with a history of excessive scarring (e.g., hypertrophic scarring and keloid formations) and pigmentation disorders has not been studied. The safety of JUVÉDERM injectable gel for use during pregnancy, in breast feeding females or in patients under 18 years has not been established.